https://www.acsh.org/news/2025/06/27/perfect-rocket-fuel-no-fires-no-chemicals-just-energy-49569
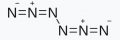
https://www.nature.com/articles/s41586-025-09032-9
According to CCSD(T)/cc-pVTZ (ΔH0) computations, the decomposition of N6 into three N2 is exothermic (ΔH0) by 185.2 kcal mol−1, which is 2.2 and 1.9 times higher than the decomposition enthalpies of TNT (2,4,6-trinitrotoluene) and HMX (1,3,5,7-tetranitro-1,3,5,7-tetrazocane, octogen) by weight50 (see the ‘Computational details’ section in Methods).
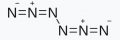
https://www.nature.com/articles/s41586-025-09032-9
According to CCSD(T)/cc-pVTZ (ΔH0) computations, the decomposition of N6 into three N2 is exothermic (ΔH0) by 185.2 kcal mol−1, which is 2.2 and 1.9 times higher than the decomposition enthalpies of TNT (2,4,6-trinitrotoluene) and HMX (1,3,5,7-tetranitro-1,3,5,7-tetrazocane, octogen) by weight50 (see the ‘Computational details’ section in Methods).

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin




