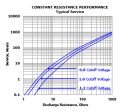
Many times I have had AA, AAA, D cell, or other standard batteries and wondered: how much current can these things supply, peak and continuous? And what is their capacity under different conditions? When making something that is intended to be low cost, for consumers, or with limited supplies, you often will use standard alkaline batteries for your design.
So what are the specs for typical AAAA, AAA, AA, C, D, and other alkaline batteries? I have some things in mind if I can get a few amps from the larger ones. This could be very useful information to someone trying to design anything using those batteries.
So what are the specs for typical AAAA, AAA, AA, C, D, and other alkaline batteries? I have some things in mind if I can get a few amps from the larger ones. This could be very useful information to someone trying to design anything using those batteries.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin