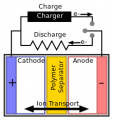
I think electricity flows through the battery because I don't understand how a battery in series that is placed between others like the one in the picture could give or receive electrons. I recently saw in a youtube video that electricity only flows through the circuit. Similiar to a capacitor it doesn't flow through it self. I took it as truth as considered it to be one of them mind blowers but it didn't seem right and after trying to disprove it in my head I thought about batteries in series. The idea of all the electrons leaving the negative for positive as well seemed like you would just mount of electrons on the positive side and run out. I'm assuming the basics of electron flow is the same in all batteries? I understand there is differences physically and in the chemistry. Thanks yall!
Attachments
-
3 KB Views: 2

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin