When you charge a battery cell, i assume electrons are entering the positive terminal to get stored in the cell. I assume, since the negative terminal is connected to ground, that ground connection is what creates current flow into the positive terminal -- without a ground connection, the cell wouldn't charge. So what flows out of the negative terminal into ground during charging? If it's electrons, doesn't that discharge the cell?
What leaves a cell's negative terminal during charging?
- Thread starter johnyradio
- Start date
Scroll to continue with content
Don't over think this problem.When you charge a battery cell, i assume electrons are entering the positive terminal to get stored in the cell. I assume, since the negative terminal is connected to ground, that ground connection is what creates current flow into the positive terminal -- without a ground connection, the cell wouldn't charge. So what flows out of the negative terminal into ground during charging? If it's electrons, doesn't that discharge the cell?
The current flowing into the battery being charged is the same as the current flowing out of the battery.
The charging source has potential energy.
Energy is transferred from the source to the battery. The potential energy of the battery rises.
I think you didn't answer the question.Don't over think this problem.
But i suspect the following:
Nothing flows out of the negative terminal during charging. Rather, it simply creates a voltage attraction, to pull electrons into the positive terminal.
So what flows out of the negative terminal into ground during charging? If it's electrons, doesn't that discharge the cell?
Absolutely not.Nothing flows out of the negative terminal during charging. Rather, it simply creates a voltage attraction, to pull electrons into the positive terminal.
For one electron to leave the battery from one terminal, one electron as to enter, since that is the only way current can flow.
Electrons don't provide the energy, they just carry it.
All the battery does is add energy due to chemical changes, which generates the battery voltage difference between its terminals, which is transferred by the electrons between the two terminals.
That sounds like a description of discharge, but charge is the same idea.For one electron to leave the battery from one terminal, one electron as to enter, since that is the only way current can flow.
According to this video, during charging, electrons leave the cathode, pass through the charger, and enter the anode.
https://youtube.com/shorts/A0gnIR5c1nY
The difference between charge and discharge is just the opposite direction of the flowing electrons.That sounds like a description of discharge, but charge is the same idea.
You assume wrong. When a battery is charging, charge flows into the anode, not electrons. Electrons are negative, so they flow in the opposite direction of charge.When you charge a battery cell, i assume electrons are entering the positive terminal to get stored in the cell.
And batteries do not store charge.
I did answer your question. You just were not paying proper attention.I think you didn't answer the question.
But i suspect the following:
Nothing flows out of the negative terminal during charging. Rather, it simply creates a voltage attraction, to pull electrons into the positive terminal.
I said current flowing in and current flowing out are the same. I did not use the word "electrons" because that makes you think of charge accumulation, which is not what is happening.
If you must, imagine you have a piece of wire connected to a battery.
An electron enters one end of the wire and another exits the other end. There is no charge accumulation on the wire.
And yet the wire gets hot. Energy is generated and dissipated as thermal energy. Thus energy is transferred, with no net gain or loss of electrons.
The same happens when the battery is charging. Electrons deliver energy with no net gain or loss of electrons in the process.
Thinking of electron flow in and out of the battery does not explain how a battery gets charged.
You didn't mention electrons because they aren't moving? Or because of what you think it makes me think? You said i shouldn't overthink. What's the correct amount to think? I'm thinking of thinking. What do you think?current flowing in and current flowing out are the same. I did not use the word "electrons" because that makes you think of charge accumulation, which is not what is happening.
This video seems to say charging is all about the transfer of ions inside the cell. If true, that helps me understand why charging is not about electron accumulation.
This article seems to agree that charging is all about the transfer of ions. Somehow, the movement of electrons from the negative to positive terminal through the charger allows or attracts the ions back toward the positive terminal.
When we charge the lithium batteries, the electrons are sent back to the anode and the lithium ions re-intercalate themselves in the cathode. This restores the battery’s capacity.
Are you speaking of ions? This video says ions travel to cathode.When a battery is charging, charge flows into the anode
Last edited:
First off, when you charge a battery, electrons leave the positive terminal and enter the negative terminal. When the battery is in use, it's the other way around.When you charge a battery cell, i assume electrons are entering the positive terminal to get stored in the cell. I assume, since the negative terminal is connected to ground, that ground connection is what creates current flow into the positive terminal -- without a ground connection, the cell wouldn't charge. So what flows out of the negative terminal into ground during charging? If it's electrons, doesn't that discharge the cell?
Forget about a battery for a moment, and think of a capacitor.
When you charge a capacitor, you move an electron from one plate and put it on the other plate (it's not the same electron, but that's irrelevant). If you then discharge the capacitor, electrons move back from the plate that has an excess to the plate that has a deficit.
Notice that the total net charge on the capacitor overall is always zero -- we have just created a separation of charges within the device.
The same is true of a battery in that, at all times, it has a net overall charge of zero. The big difference is that instead of creating a physical separation of charges like we do in a capacitor, we are controlling chemical reactions that store or release energy in the chemical bonds that are created and/or broken. The amount of actual charge separation that exists within a battery is miniscule.
Thx, i'm getting that.when you charge a battery, electrons leave the positive terminal and enter the negative terminal. When the battery is in use, it's the other way around. it has a net overall charge of zero.
Is the following correct:
The movement of the ions during discharge causes movement of electrons between terminals (through the load), which in turn cause electromagnetic motion in the surrounding wires, which is where the real power (energy?) is.
At the moment, just trying to grasp cell charging. EM fields was a tangent (get it?If you want to get into where the energy really is, then you need to be talking about the electric and magnetic fields, since that is where the energy resides. But that may or may not be useful depending on what you are actually trying to grasp.
Ions do not flow in or out of the battery. They do flow inside the battery. But that has nothing to do with your question.Are you speaking of ions? This video says ions travel to cathode.
It might be a little related.Ions do not flow in or out of the battery. They do flow inside the battery. But that has nothing to do with your question.
Yes, i understand the ions remain inside the cell.
Here is a thought experiment for you.
In this real working circuit, AC voltage is applied to the primary winding of transformer T1.
On the secondary side, battery BT1 becomes charged even though there are no electrons passing from the primary to the secondary winding.
Capacitor C1 is not necessary but just added to emphasize that there is no DC flowing into the transformer.
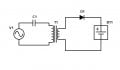
In this real working circuit, AC voltage is applied to the primary winding of transformer T1.
On the secondary side, battery BT1 becomes charged even though there are no electrons passing from the primary to the secondary winding.
Capacitor C1 is not necessary but just added to emphasize that there is no DC flowing into the transformer.

Transformers are magic
There's zero magic in imaginary toys.
Electricity is real magic.
The toys are very real -- certainly people pay real money for them.There's zero magic in imaginary toys.
Well, perhaps to a lay person.Electricity is real magic.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin