Hello.
I'm kind of conducting a very simple study at work on heat transfer to try to compare 2 different heating systems of enclosed spaces but I'm having a bit of a struggle to understand the meaning of some of the values I'm getting and I'm seeking some help to see if the values makes sense in the context!
So, the idea is to use 2 formulas (Q = m*C*ΔT and Qloss = A * ΔT / Rt) to calculate energy needed to heat up a place and to calculate the heat losses so that, in the end of the day I can look to the values and say something like:
- with system A1 I need X1 amount of energy and it will cost me Y1€ to run it over a given period of time.
- with system A2 I need X2 amount of energy and it will cost me Y2€ to run it over the same period of time.
I've been working on this using a spreadsheet of course.
So, to start I need to give some context.
I'm running these formulas to calculate energy needed to heat up a room which I'm going to assume the following properties:
A is area = 101.49m² (ignoring the floor)
V is volume = 97.89m³ (ignoring the floor)
Then, the room wall is composed of 2 walls of plasterboard and an air gap. Dimensions are as follows:
dp is thickness of plasterboard = 0.01m (x2 walls of it)
da is thickness of air = 0.04m
To calculate energy needed to heat this space up, I have considered two different temperature targets to account to colder and hotter time frames.
My target temperature is Tin, which is the temperature I want to keep constant inside the enclosed space.
Tout1/2 is the ambient temperature which I am considering two fo them, so this will give me 2 different ΔT, ΔT1 and ΔT2.
So:
Tin = 17ºC
Tout1 = 3.8ºC
Tout2 = 10.4ºC
ΔT1 = 13.2ºC
ΔT2 = 6.6ºC
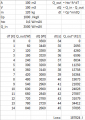
So, I get the following results:

What I'm not sure if I'm doing correctly is for the heat losses. The formula I'm using gives me Watts [W] or Joules per second [J/s] which is a rate. So, to calculate the heat losses over a day, I should simply multiply the results of Qloss by 86400 seconds which is the number of seconds of a day and finally add that value to the heat needed to initially heat up the space?
I'm kind of conducting a very simple study at work on heat transfer to try to compare 2 different heating systems of enclosed spaces but I'm having a bit of a struggle to understand the meaning of some of the values I'm getting and I'm seeking some help to see if the values makes sense in the context!
So, the idea is to use 2 formulas (Q = m*C*ΔT and Qloss = A * ΔT / Rt) to calculate energy needed to heat up a place and to calculate the heat losses so that, in the end of the day I can look to the values and say something like:
- with system A1 I need X1 amount of energy and it will cost me Y1€ to run it over a given period of time.
- with system A2 I need X2 amount of energy and it will cost me Y2€ to run it over the same period of time.
I've been working on this using a spreadsheet of course.
So, to start I need to give some context.
I'm running these formulas to calculate energy needed to heat up a room which I'm going to assume the following properties:
A is area = 101.49m² (ignoring the floor)
V is volume = 97.89m³ (ignoring the floor)
Then, the room wall is composed of 2 walls of plasterboard and an air gap. Dimensions are as follows:
dp is thickness of plasterboard = 0.01m (x2 walls of it)
da is thickness of air = 0.04m
To calculate energy needed to heat this space up, I have considered two different temperature targets to account to colder and hotter time frames.
My target temperature is Tin, which is the temperature I want to keep constant inside the enclosed space.
Tout1/2 is the ambient temperature which I am considering two fo them, so this will give me 2 different ΔT, ΔT1 and ΔT2.
So:
Tin = 17ºC
Tout1 = 3.8ºC
Tout2 = 10.4ºC
ΔT1 = 13.2ºC
ΔT2 = 6.6ºC
So, I get the following results:

What I'm not sure if I'm doing correctly is for the heat losses. The formula I'm using gives me Watts [W] or Joules per second [J/s] which is a rate. So, to calculate the heat losses over a day, I should simply multiply the results of Qloss by 86400 seconds which is the number of seconds of a day and finally add that value to the heat needed to initially heat up the space?

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin