Calculate the energy of the Photon
- Thread starter Mojo Pin__
- Start date
Scroll to continue with content
AlbertHall
- Joined Jun 4, 2014
- 12,624
For the n=1 to n=3 transition the photon must supply the difference in energy between the two levels.
For the second case the final energy is just the kinetic energy as the electron is not part of the atom.
For the second case the final energy is just the kinetic energy as the electron is not part of the atom.
For the n=1 to n=3 transition the photon must supply the difference in energy between the two levels.
For the second case the final energy is just the kinetic energy as the electron is not part of the atom.
Hi! Thanks for the response
For transition 2 I thought that because the electron starts at energy level 3 and then is ejected there is only:
1/nlow^2
because there is no higher energy level I ignore the:
-1/nhigh^2
but include kinetic energy to calculate the energy of photon 2.
Do you think this would be an acceptable way of thinking about it?
Cheers!
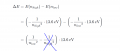

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin