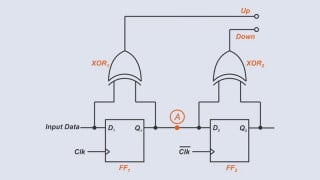
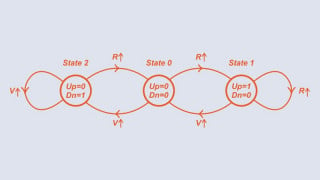
How is this determined?
Does the internal resistance change as a battery accepts a charge? It would appear so since the current flow starts to diminish once a power source is supplying current to the battery but...
As a battery is charged up its voltage rises. To what extent does the voltage rise in the battery being charged affect the current flow?
If the internal resistance rises as the battery charges (reducing current flow) and if the battery voltage rising reduces the current flow, how does one know the extent to which is doing what?
Thanks in advance for any replies.
Does the internal resistance change as a battery accepts a charge? It would appear so since the current flow starts to diminish once a power source is supplying current to the battery but...
As a battery is charged up its voltage rises. To what extent does the voltage rise in the battery being charged affect the current flow?
If the internal resistance rises as the battery charges (reducing current flow) and if the battery voltage rising reduces the current flow, how does one know the extent to which is doing what?
Thanks in advance for any replies.

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin