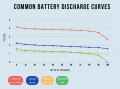
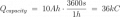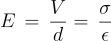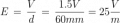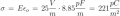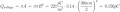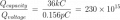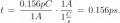I just had an interesting conversation with the neighbor who was doing some minor electrical work on his vehicle. He was wondering why a voltage reading at the positive terminal drops when a battery is connected to a (resistive) load. I told him that the battery is sort of like a large capacitor connected in series with a resistor, and thus the load forms a voltage-divider with the battery's "internal resistor". Makes sense, he said, but then he raised the point that a capacitor discharges linearly whereas the battery discharge curve is generally not so much. To that I could only say that it was basically due to the fact that the chemistry of the battery is such that the charge throughout it is roughly uniform. It was a rather hand-wavy explanation, I admit, but I honestly couldn't think of a better way to describe what is really going on there.
Is there a more concise way to explain that (and moreover, was my capacitor-resistor analogy somewhat reasonable)?
Is there a more concise way to explain that (and moreover, was my capacitor-resistor analogy somewhat reasonable)?

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin