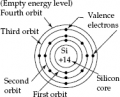
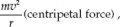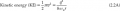So it is well known that the third valence shell of an atom can contain 18 electrons max.
However, in a silicon atom, the outermost shell is considered to be full with only 8 electrons.
Why is this?
It has something to do with energy level groups but i dont understand it.
I found this but still dont get it
Does the 3rd electron shell have a capacity for 8e- or 18e-? | Socratic
Can someone explain?
Thanks
However, in a silicon atom, the outermost shell is considered to be full with only 8 electrons.
Why is this?
It has something to do with energy level groups but i dont understand it.
I found this but still dont get it
Does the 3rd electron shell have a capacity for 8e- or 18e-? | Socratic
Can someone explain?
Thanks