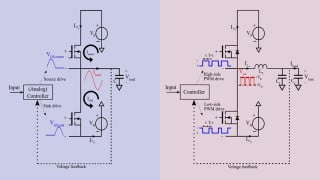
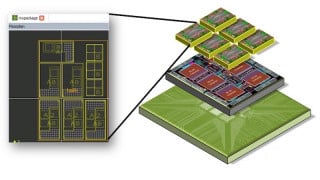
Greetings,
I've been diving into the world of soldering and flux recently, and I'd like to check my comprehension with those more experienced in this field. This is my current understanding, and I would greatly appreciate your input if there are any misconceptions or omitted parts:
During soldering, we heat up metals that can oxidize when they come into contact with air. The oxidized layer that forms can hinder the solder's ability to properly bond with the metal, which is where the role of flux becomes critical.
My understanding is that flux possesses reducing properties. It acts to remove existing oxides and prevents the formation of new ones during the soldering process, ensuring a strong bond between the solder and the metal.
However, the residues that the flux leaves behind can cause corrosion over time. This becomes a specific issue in electronics, as the corrosion can lead to increased electrical resistance, formation of unintentional conductive paths (which could cause short circuits), or even result in complete component failure.
Therefore, in the realm of electronics, it becomes necessary to use non-corrosive fluxes, such as rosin flux, to avoid these problems. I also understand that there are other types of flux, like acid core flux and plumber's flux, which are corrosive and thus unsuitable for electronics.
Is my understanding accurate? Are there any areas that I've misunderstood or overlooked? Any clarification or additional insights would be hugely beneficial!
I've been diving into the world of soldering and flux recently, and I'd like to check my comprehension with those more experienced in this field. This is my current understanding, and I would greatly appreciate your input if there are any misconceptions or omitted parts:
During soldering, we heat up metals that can oxidize when they come into contact with air. The oxidized layer that forms can hinder the solder's ability to properly bond with the metal, which is where the role of flux becomes critical.
My understanding is that flux possesses reducing properties. It acts to remove existing oxides and prevents the formation of new ones during the soldering process, ensuring a strong bond between the solder and the metal.
However, the residues that the flux leaves behind can cause corrosion over time. This becomes a specific issue in electronics, as the corrosion can lead to increased electrical resistance, formation of unintentional conductive paths (which could cause short circuits), or even result in complete component failure.
Therefore, in the realm of electronics, it becomes necessary to use non-corrosive fluxes, such as rosin flux, to avoid these problems. I also understand that there are other types of flux, like acid core flux and plumber's flux, which are corrosive and thus unsuitable for electronics.
Is my understanding accurate? Are there any areas that I've misunderstood or overlooked? Any clarification or additional insights would be hugely beneficial!

 Facebook
Facebook Google
Google GitHub
GitHub Linkedin
Linkedin